An Optimized Marinopyrrole A Derivative Targets 6‑Phosphoglucosamine Synthetase to Inhibit Methicillin-Resistant Staphylococcus aureus
Fusheng Guo, Fan Xiao, Hao Song, Xiaoyong Li, Yaxin Xiao, Yong Qin,* and Xiaoguang Lei*
ACS central science, 2024. DOI: 10.1021/acscentsci.4c01167
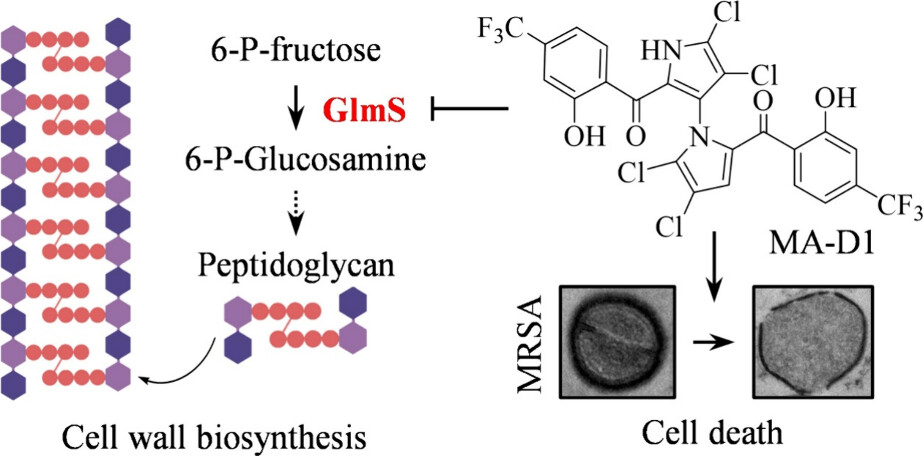
Methicillin-resistant Staphylococcus aureus (MRSA) is a common pathogenic bacterium that causes clinical infection and has become one of the most prominent antibiotic-resistant bacteria in the world. There is a pressing need to develop new antibiotics based on novel modes of action to combat increasingly severe MRSA infection. Marinopyrrole A (MA), a natural product extracted from marine Streptomyces in 2008, has a unique bipyrrole chemical skeleton and shows potent antibacterial activity against MRSA. However, its mode of action is still elusive. Herein, we developed an optimized MA derivative, MA-D1, and applied a chemoproteomic approach to reveal that MA-D1 performs its anti-MRSA activity by directly targeting 6-phosphoglucosamine synthetase (GlmS) to cause the breakdown of bacterial cell wall biosynthesis. Computational and experimental studies showed that MA-D1 interacts with the key R381 and E382 residues of GlmS in a novel binding pocket. Furthermore, MA-D1 showed a low resistance frequency for MRSA treatment and was also sensitive against the linezolid-, vancomycin-, or teicoplanin-resistant MRSA strains. MA-D1 also showed in vivo antibiotic efficacy in multiple animal models. This study demonstrates the promising potential of targeting GlmS to develop a new class of antibiotics to control MRSA pathogen infection.